\[p_1 = \frac {e^{-E_1/k_BT}}{e^{-E_1/k_BT}+e^{-E_2/k_BT}} = \frac {e^{\epsilon /k_BT}}{e^{\epsilon /k_BT}+e^{-3\epsilon /k_BT}} = \frac {e^{2\epsilon /k_BT}}{e^{2\epsilon /k_BT}+e^{-2\epsilon /k_BT}}\]
\[p_2 = \frac {e^{-E_2/k_BT}}{e^{-E_1/k_BT}+e^{-E_2/k_BT}} = \frac {e^{-3\epsilon /k_BT}}{e^{\epsilon /k_BT}+e^{-3\epsilon /k_BT}} = \frac {e^{-2\epsilon /k_BT}}{e^{2\epsilon /k_BT}+e^{-2\epsilon /k_BT}}\]
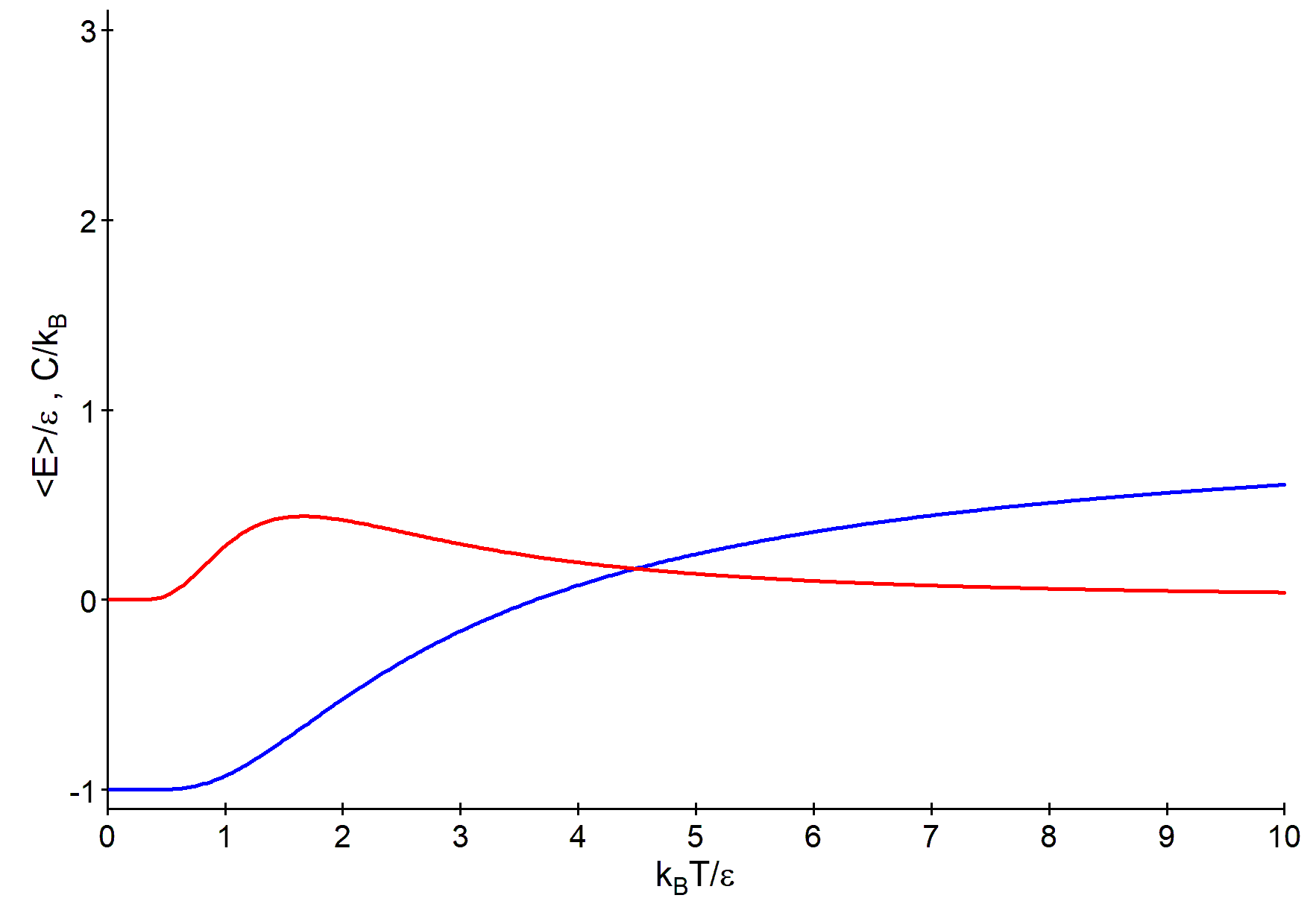
\(T\to 0\): \(\qquad \epsilon /k_BT \to \infty \qquad \Rightarrow \langle E \rangle = \epsilon - 2\epsilon \tanh (\infty ) = -\epsilon \)
\(T\to \infty \): \(\qquad \epsilon /k_BT \to 0 \qquad \Rightarrow \langle E \rangle = \epsilon - 2\epsilon \tanh (0) = \epsilon \)
\[ C = \frac d {dT} \langle E \rangle = -2\epsilon \frac d {dT} \tanh (2\epsilon /k_B T) -2 \epsilon \frac 1 {\cosh ^2(2\epsilon /k_B T)} \frac {-2\epsilon }{k_B T^2} = k_B \left ( \frac {2\epsilon }{k_B T}\right )^2 \frac 1 {\cosh ^2(2\epsilon /k_B T)} \]
\(T\to 0\): \(\qquad \epsilon /k_BT \to \infty ~, \qquad x^2/\cosh ^2(x) \to 0 \) for \(x\to \infty ~, \qquad \Rightarrow C=0\)
\(T\to \infty \): \(\qquad \epsilon /k_BT \to 0 ~, \qquad x^2/\cosh ^2(x) \to 0 \) for \(x\to 0 ~, \qquad \Rightarrow C=0\)
Find maximum of \(f(x)= x^2/\cosh ^2(x)\):
numerical solution: \(x_{max} = 1.1997 \qquad k_B T_{max} = 2\epsilon / 1.1997 = 1.667 \epsilon \)