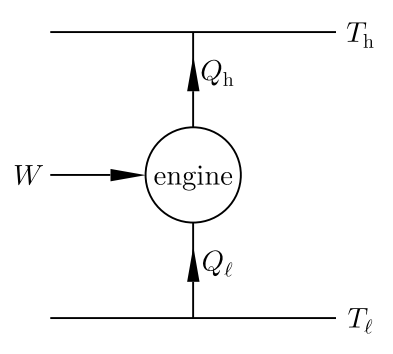
A heat pump is a device that uses work to transport heat from a low-temperature reservoir to a high-temperature reservoir (see figure on the right). Its efficiency can be defined as the ratio of the heat deposited into the high-temperature reservoir and the work done on the system, .
Heat pumps can be used to heat buildings. What is the efficiency of an ideal (Carnot) heat pump that takes heat from the outside air at 40 F and transports it to the inside of the building which is at 72 F?
What is the efficiency of the heat pump in part c) if the outside air is at a temperature of 0 F?

An ideal air conditioner consists of a Carnot cycle (running backwards). It absorbs heat from the inside of a house at the lower temperature and discharges heat to the outside at the higher temperature , consuming electric energy . The heat leaking back into the house through the walls and windows is given by where is a constant.
The air conditioner runs continuously, and the temperature in the house has reached a steady state. Derive a relation for the inside temperature in terms of , , and .
The system is designed such that it runs at half of the maximum electrical energy input if the outside temperature is 90 F and the inside temperature is 72 F. What is the highest outside temperature for which the system can maintain an inside temperature of 72 F at full electrical input.
Consider a paramagnetic substance whose equation of state reads where is the temperature, is the magnetization, is the magnetic field, and is a material specific constant. The internal energy is given by where the specific heat is a constant. The work differential for a paramagnet is
Consider an isothermal change of magnetization from to . Compute the work done on the system and the heat absorbed by the system.
Consider an adiabatic change of magnetization from to . Find the adiabatic curves by starting from and integrating the resulting differential equation.
Sketch a Carnot cycle, consisting of two isothermal changes in and two adiabatic changes in in the plane.
Compute the total work during the cycle and the heat absorbed during the four segments of the cycle.
Explicitly calculate the efficiency.
Hint: The derivation is analogous to that of the Carnot cycle for the ideal gas, but using the equation of state instead of the ideal gas law. This leads to changes in some of the mathematical expressions.