A Diesel engine takes in air at ambient conditions (room temperature, 1 atm pressure) and compresses it rapidly to about 5% of its original volume.
Calculate the temperature of the air at the end of the compression. Assume that the compression is so fast that it can be assumed to be adiabatic. The adiabatic index for air is about 1.4.
Explain why a Diesel engine does not need spark plugs.
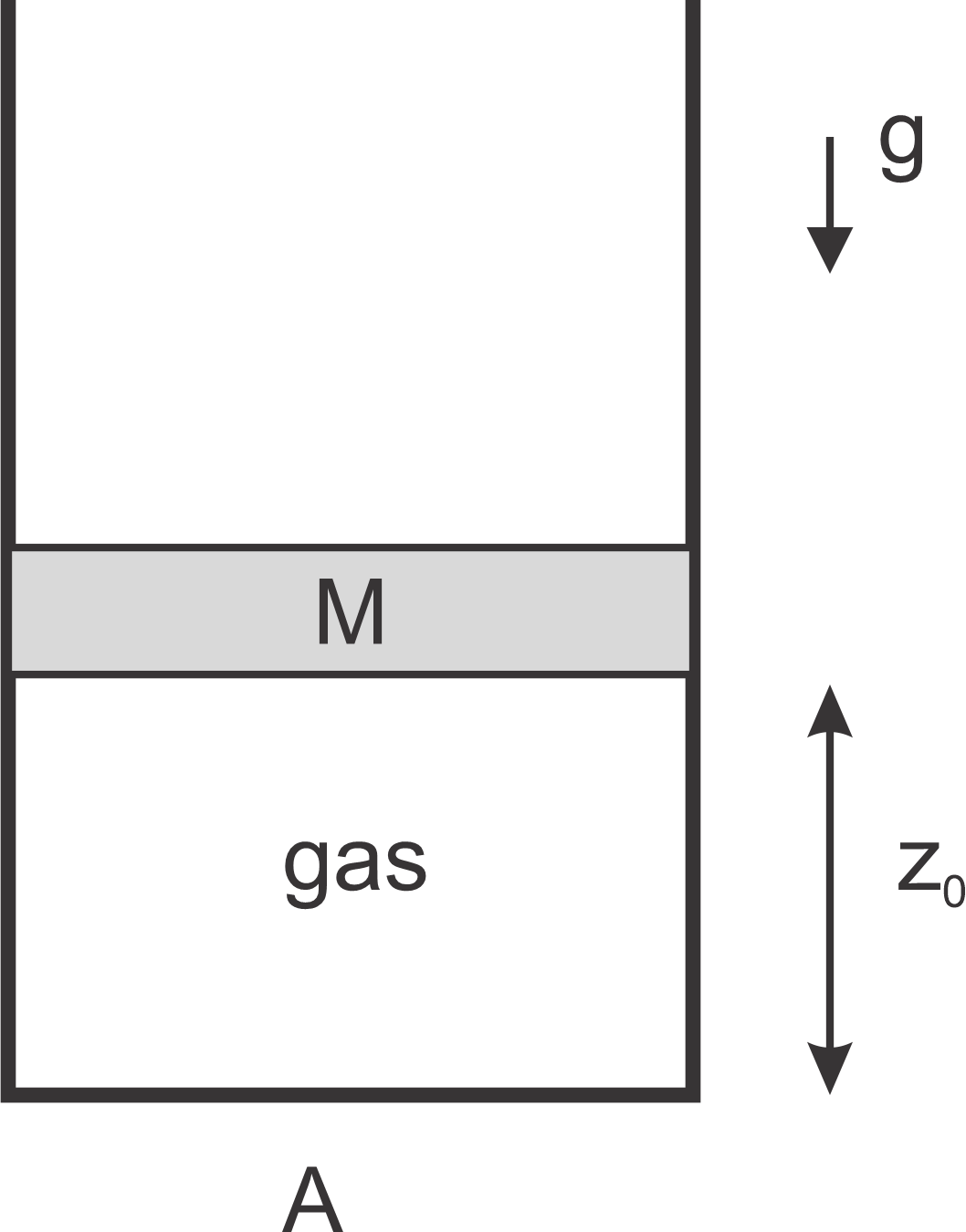
An ideal gas of
atoms is contained in a cylindrical vessel of cross section
with a piston
of mass
on top. The piston can move up and down, keeping the pressure constant,
but the gas cannot escape. The device is surrounded by vacuum.
Initially, the cylinder is at rest, the gas is in equilibrium at temperature
, and the vertical position
of the cylinder is .
Consider the forces acting on the piston, and find the initial pressure inside the gas.
The gas is now heated up slowly, lifting the piston. Find the temperature of the gas, when the height of the cylinder reaches .
Compute the work done by the gas during the expansion. (Does the pressure change in this process?)
Find the change in the internal energy of the gas in this process.
How much heat has to be provided to the gas in this process? Express the answer in terms of and .

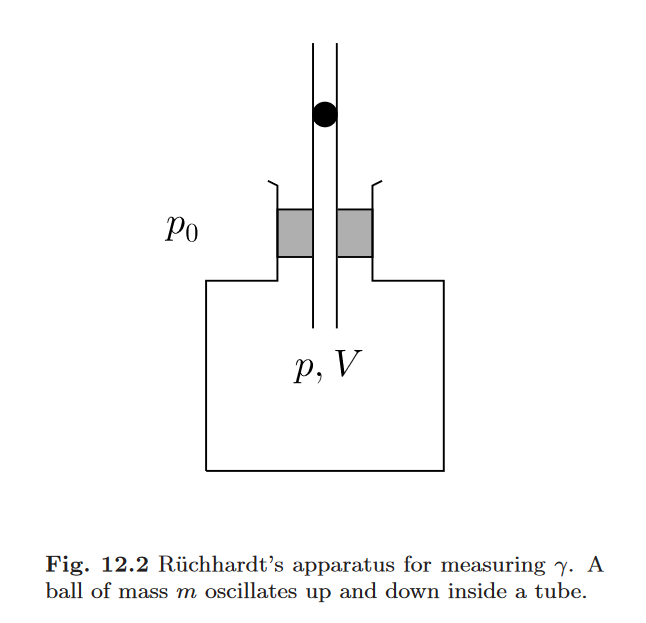
Consider the device shown in the figure: A ball of mass
is placed snugly in a tube of cross section
connected to a container containing an ideal gas. The ball
can move up and down, but gas cannot escape. In the
equilibrium position of the ball, the enclosed gas volume is
.
The device is surrounded by air at ambient pressure
.
Consider the forces acting on the ball, and find the pressure inside the gas if the ball is at rest in its equilibrium position.
The ball is now given a small downward displacement from its equilibrium position. Compute the force acting on the ball. (Assume that the displacement is fast enough so that the compression of the gas can be treated as adiabatic because no heat is exchanged with the environment.)
The ball is now released. Determine the period of its oscillations. (Assume that the system remains thermally isolated during the oscillations. Also neglect friction.)