Consider a system of independent atoms, each having three states with energies , . The system is in equilibrium with a heat bath at temperature .
Calculate the partition function of a single atom and the partition function of the -atom system.
Find the Helmholtz free energy and the internal energy.
Calculate the entropy and discuss its behavior for . Where does the excess entropy come from?
Consider a classical degree of freedom that makes a contribution to the Hamiltonian of the form where and are positive constants. Find the average internal energy stored in this degree of freedom as a function of temperature.
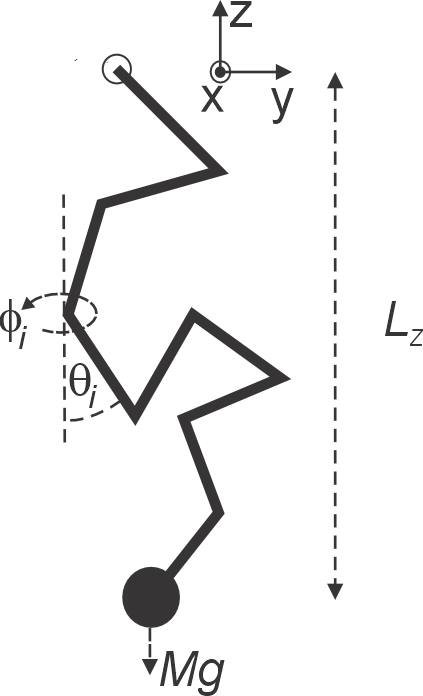
A model of a polymer consists of a large number of identical rods (“monomers”) of length , attached end to end as shown in the figure. The connections between the rods are completely flexible so that the orientation of each rod (characterized by the spherical angles and w.r.t. the -axis) is independent of the neighboring rods. One end of the polymer is fixed, the other end is attached to a weight of mass . Neglecting the mass of the monomers, the energy of a given state of the polymer can be written as
The spherical angles take values and . The system is in thermal equilibrium at temperature .

Calculate the single-monomer partition function .
Find the partition function of the entire polymer and its free energy.
Compute the average vertical distance between the fixed end of the polymer and the mass as a function of , , , and .
Analyze your result for in the limits of and . Compare the resulting values of to your physical expectation.
At high temperatures, , the polymer acts like a spring, i.e., there is a linear relation between between the applied force and its vertical length . Find this relation from your result in part c). [Hint: Taylor expansion of to the lowest non-vanishing order]